Improved H2O2 photogeneration by KOH-doped g-C3N4 under visible light irradiation due to synergistic effect of N defects and K modification
Hao Zhang, Luhan Jia, Pan Wu, Rongjie Xu, Jian He, Wei Jiang⁎
Cite this: Applied Surface Science 527 (2020) 146584
https://doi.org/10.1016/j.apsusc.2020.146584
Abstract:
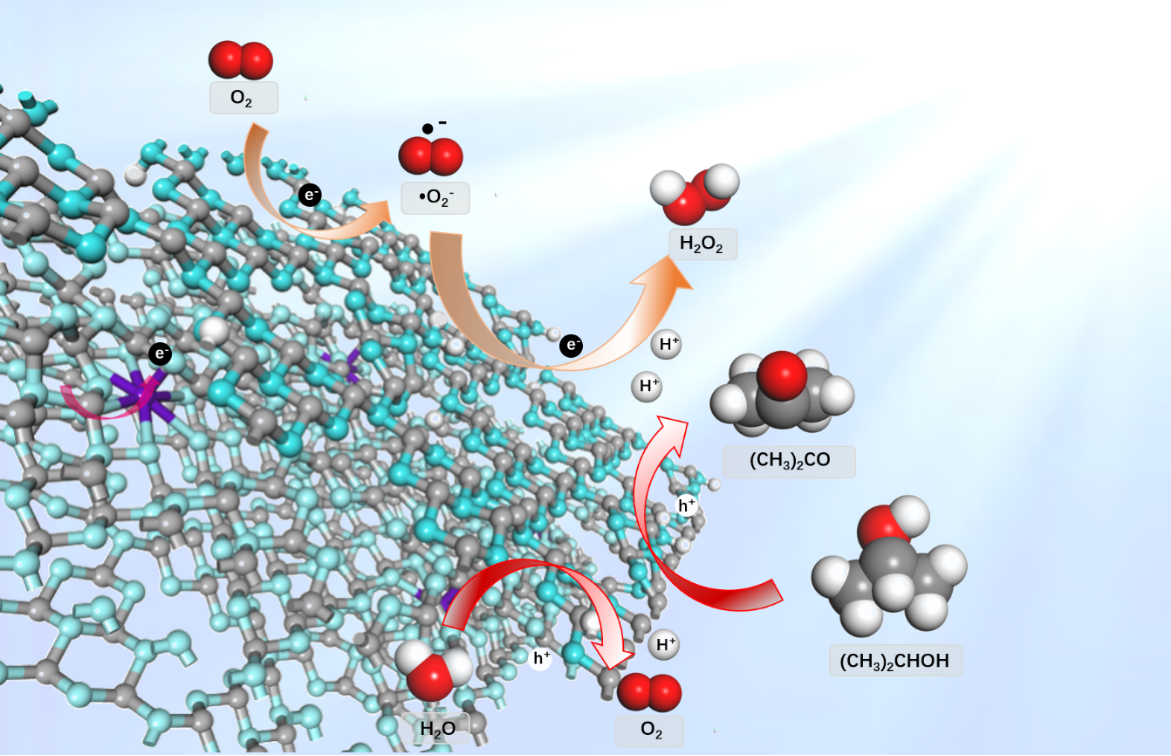
KOH-doped graphitic carbon nitride (g-C3N4) prepared by thermal copolymerization in the presence of KOH was used for photogeneration of H2O2 under visible light irradiation. The H2O2 yield of the KOH-doped g-C3N4 photocatalyst with isopropanol (IPA) as a scavenger was 704 μM·g−1 g-C3N4·h−1, twelve times that of unmodified gC3N4. The total H2O2 output with IPA as a scavenger increased to 86.08 mM after 60 h. The photogeneration of H2O2 with g-C3N4 occurred as two single-electron reactions in which oxygen was reduced to the superoxide radical by one photogenerated electron and further reduced to H2O2 by reacting with another electron and two protons released by oxidization of IPA by holes. KOH doping had a synergistic effect owing to the strong electron-withdrawing ability of N defects caused by N vacancies and cyano groups, and enhanced electron transport due to K bridges between the C3N4 layers. These effects significantly enhanced the H2O2 photogeneration performance of g-C3N4 by improving the quantum efficiency, narrowing the band gap, strengthening IPA adsorption on g-C3N4, and lowering the energy barrier of the oxygen reduction reaction. This work provides an attractive and promising strategy for green H2O2 production.
该工作通过简单的热缩和法制备了富含N缺陷的K掺杂氮化碳,N缺陷和K掺杂的协同作用抑制了电子和空穴的复合频率,提高了量子效率。通过DFT计算,经N缺陷和K掺杂修饰后,降低了可见光下氧气的两步单电子还原的能垒,进一步提高了H2O2的产量。为光催化生产情清洁能源提供了基础。
KBiO3 as an Effective Visible-Light-Driven Photocatalyst: Stability Improvement by In Situ Constructing KBiO3/BiOX (X = Cl, Br, I) Heterostructure
Hao Zhang, Hanxiao Zheng, Ying Wang, Runhua Yan, Dingyuan Luo, and Wei Jiang*
Cite this: Ind. Eng. Chem. Res. 2019, 58, 1875−1887.
https://pubs.acs.org/doi/10.1021/acs.iecr.8b05658
Abstract:
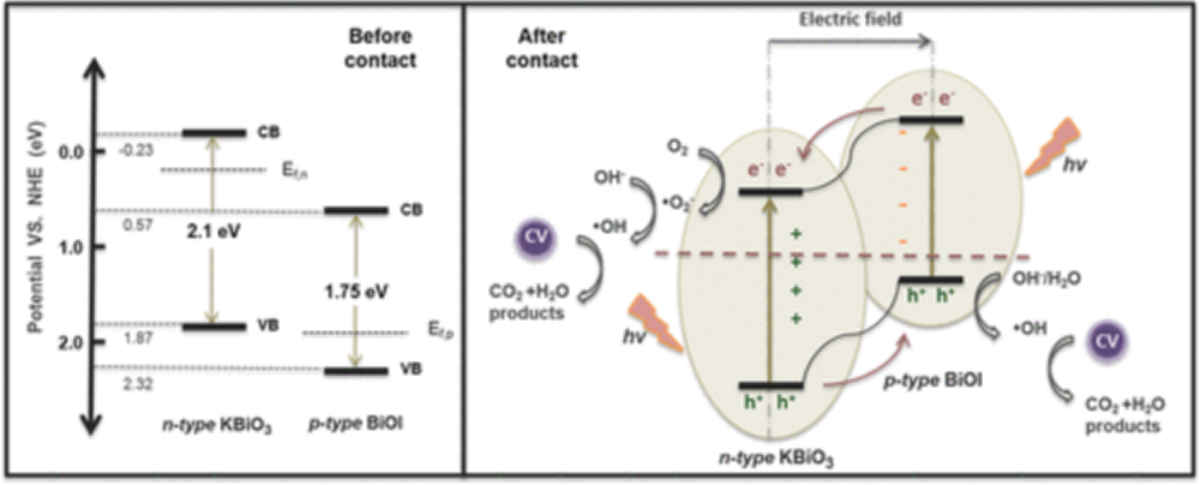
Petal bismuth salt semiconductors are promising as visible-light-driven photocatalysts, but their short service times due to strong oxidation limit their application. In this research, the in situ construction of a p−n heterojunction, by growing BiOX on a KBiO3 surface, is used as a strategy to improve the performance and stability. Evaluation of the photocatalytic degradation of crystal violet and phenol shows that the KBiO3/BiOX (X = Cl, Br, I) heterostructures exhibit significant improvement in photocatalytic performance and stability compared to KBiO3. The best one, KBiO3/(5.85%) BiOI, shows 98% degradation of crystal violet within 20 min, and of phenol within 1.5 h. No significant performance decay can be detected after repeated use. The development of KBiO3/BiOX heterostructure improves the applicability of KBiO3 as an efficient visible-lightdriven photocatalyst.
本研究采用原位生成策略合成了一系列KBiO3/BiOX (X = Cl, Br, I)复合材料。KBiO3和BiOX之间形成异质结结构得到的异质结KBiO3/BiOX复合材料光催化性能有明显改善,且随卤素原子序数的增加,其性能增强程度增大。以KBiO3/(5.85%) BiOI为最佳复合材料,可在20 min和1.5 h内有效地光降解CV和苯酚。KBiO3带隙较窄,可以作为一种优秀的可见光驱动光催化剂,与BiOX形成p-n异质结可以进一步提高其活性和稳定性。
Magnetic MFe2O4-Ag2O (M = Zn, Co, & Ni) composite photocatalysts and their application for dye wastewater treatment
Fujin Sun, Qihui Zeng, Wen Tian, Yingming Zhu, Wei Jiang*
Cite this: Journal of Environmental Chem. Eng., Volume 7, Issue 2, April 2019, 103011
https://doi.org/10.1016/j.jece.2019.103011

Abstract:
Silver oxide is a high-performance visible-light-driven nanoparticle photocatalyst. However, its high cost and difficult recovery limit its wide application. Constructing a magnetic composite is a smart strategy to effectively utilize nanoparticle photocatalysts with acceptable performance maintenance. In this study, three magnetic ferrite supports, that is, CoFe2O4, ZnFe2O4, and NiFe2O4, were used as support to load Ag2O and construct Ag2O/MFe2O4 composites to lower the Ag consumption and intensify the recovery rate. The photoactivity, stability, and magnetic recovery of the three composite magnetic photocatalysts were evaluated by using four typical organics. The results of the batch experiment confirm that Ag2O/MFe2O4 (60%) possesses a good photoactivity and stability. However, Ag2O/ZnFe2O4 exhibits a better photoactivity and higher magnetic recovery based on cycling during the semi-continuous process. The ferrites are inert for photocatalysis process without significant effect on Ag2O photocatalytic performance, but increase the surface area and improve the recovery rate as magnetic composite carriers.
磁性复合材料是利用纳米颗粒光催化剂对废水进行分离和回收的一种智能策略。氧化银是一种高性能的可见光驱动的纳米颗粒光催化剂。但其成本高、回收难度大,限制了其广泛应用。本研究以CoFe2O4、ZnFe2O4、NiFe2O4三种磁性铁氧体载体作为载体,负载Ag2O,构建Ag2O/MFe2O4复合材料,降低Ag2O的消耗,提高了回收率。用四种典型的有机化合物对三种复合磁性光催化剂的光活度、稳定性和回收率进行了评价。间歇实验结果证实了Ag2O/MFe2O4具有良好的光活性和稳定性。然而,在半连续过程中,Ag2O/ZnFe2O4表现出更好的光活性和更高的磁回收。为磁性催化剂的进一步应用奠定了基础。
Preparation and application of separable magnetic Fe3O4-SiO2-APTES-Ag2O composite particles with high visible light photocatalytic performance
Wei Jiang*, Fujin Sun,Ya Zeng, Qihui Zeng, Tao Zhang, Wen Tian,Bin Liang
Cite this: Journal of Environmental Chem. Eng., Volume 6, Issue 1, February 2018, Pages 945-954
https://doi.org/10.1016/j.jece.2018.01.022
Abstract:
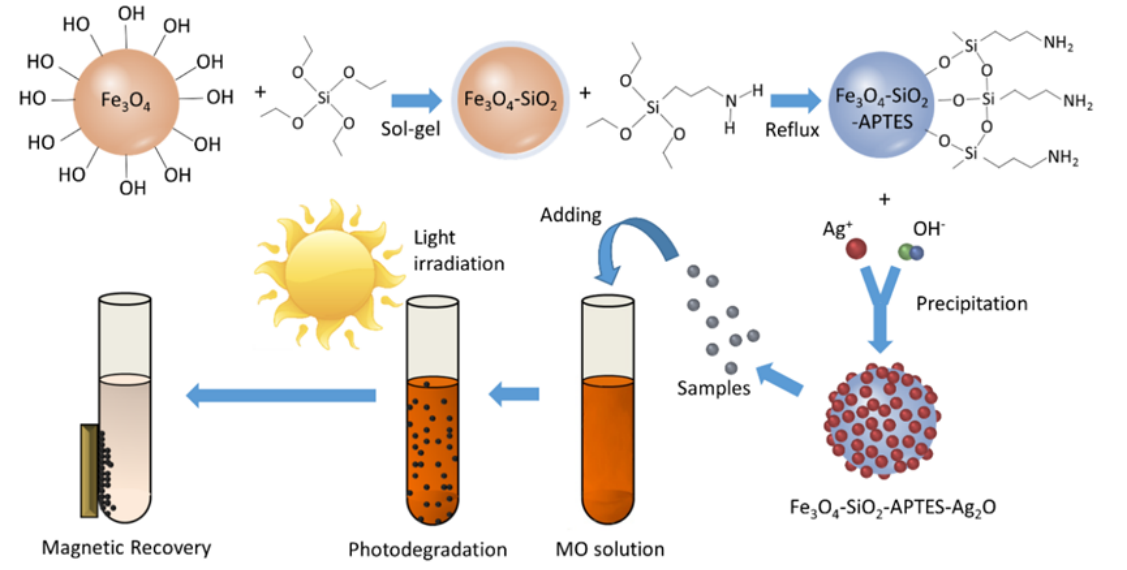
Silver oxide is a potential visible-light driven photocatalyst for the degradation of organic pollutants, but its high cost and energy intensive recovery limit its future applications. In this study, a magnetic core-shell composite, Fe3O4-SiO2-APTES-Ag2O, was prepared and evaluated. Results show that the obtained composite possesses equivalent photocatalytic performance to the pure Ag2O under visible light irradiation. Phenol and methyl orange can be effectively photodegraded with the composite catalyst in 10 min. The used composite can be efficiently separated with greater than a 98.95% recovery rate in 5 s with a 330 mT magnetic field after four cycles. In addition, the composite only exhibited negligible performance decay due to the slight loss of loaded Ag2O. Evaluations of the Fe3O4-SiO2-APTES-Ag2O composite performance in a batch reactor, a semi-continuous reactor, and a continuous magnetic agglomeration reactor with methyl orange, all confirm the superb performance, stability, and separability of this magnetic composite photocatalyst.
氧化银是一种潜在的可见光驱动的光催化剂,用于降解有机污染物,但其高成本和能源密集型回收限制了其未来的应用。本研究制备了一种磁性核-壳复合材料Fe3O4-SiO2-APTES-Ag2O,并对其进行了评价。结果表明,该复合材料在可见光照射下有相当优异的光催化性能。并将其在间歇反应器、半连续反应器和连续磁凝聚反应器中性能进行了评价,均证实了该磁性复合光催化剂的优异性能、稳定性和可分离性。
Preparation of Superhydrophobic Cu Mesh and Its Application in Rolling-Spheronization Granulation
Wei Jiang,* Jian He, Ming Mao, Shaojun Yuan, Houfang Lu, and Bin Liang
Cite this: Ind. Eng. Chem. Res. 2016, 55, 19, 5545-5555 ISSN: 0888-5885
https://doi.org/10.1021/acs.iecr.5b04685
Abstract:
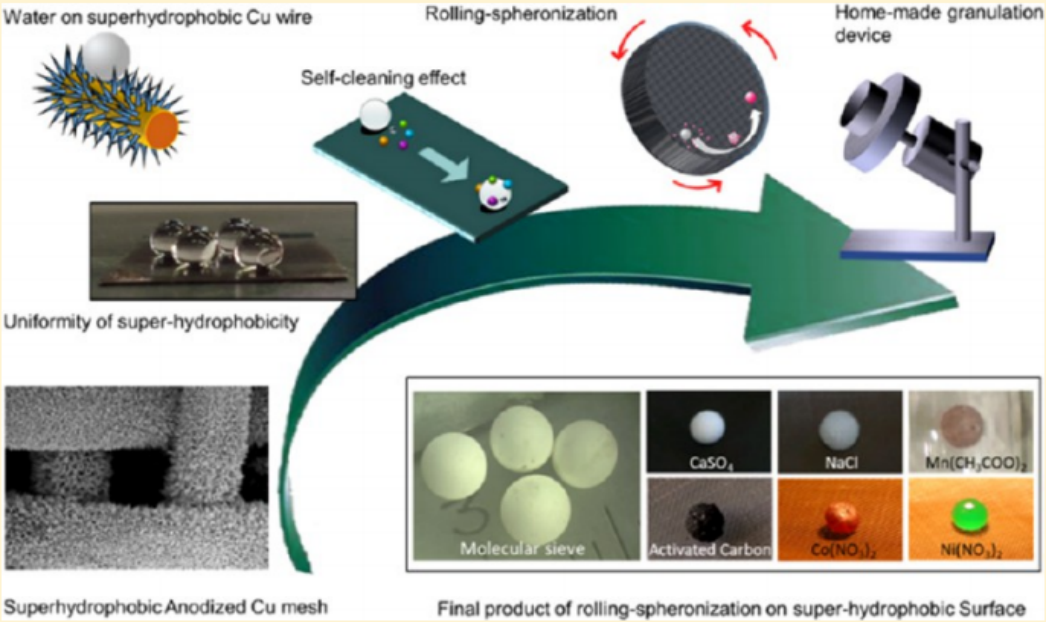
In this research, an experiment was conducted by introducing superhydrophobic surface into rolling spheronization granulation. The employed superhydrophobic surface was prepared with modification of an anodized Cu mesh with silane FAS-17 and exhibited excellent uniformity, adequate stability, and broad adaptability in the granulation scenario. Completely spherical molecular sieve granules with 99.85% sphericity and 2.53 mm diameter were obtained. The compressive strength of a single granule reached 7.402 N/ea. Only negligible residual mass and slight surface abrasion were observed in the granulation process. Force analysis confirmed that the staged motion behavior of droplets caused by the interaction between the slurry and the superhydrophobic surface benefited to the formation of spherical granules. Successful application of this process for granulation of other substances confirmed its wide suitability. With the advantages of easy fabrication, high-quality granules, and low cost, rolling-spheronization granulation on superhydrophobic surfaces has great potential for scale-up applications.
本工作采用铜网电化学阳极氧化与FAS-17氟化反应相结合的方法,制备了一种超疏水性可控的铜网,由此得到的铜网用作滚圆造粒的托盘。研究了在超疏水表面滚珠造粒的可行性和原理,并对产品进行了表征,研究的目的是评价滚珠造粒在自制超疏水表面。其中,通过在经FAS-17修饰的阳极铜网片上制备分子筛球,在超疏水表面进行滚圆法制粒,完全球形颗粒的平均球形度可达99.85%,直径为2.53 mm,抗压强度可达7.402 N/ ea,表现出良好的均匀性、足够的稳定性和广泛的适应性。在制粒过程中,仅观察到少量的超疏水表面残留质量和轻微的磨损。
First Principles Study on Formation Mechanism of Anodization Process of Titanium
Ming Mao, Ruocheng Han, Rong Zhao, Wei Jiang*, and Bin Liang
Cite this: Protection of Metals and Physical Chemistry of Surfaces, 2016, Vol. 52, No. 3, pp. 500–511. ISSN: 2070-2051
sci-hub.tw/10.1134/s2070205116030199
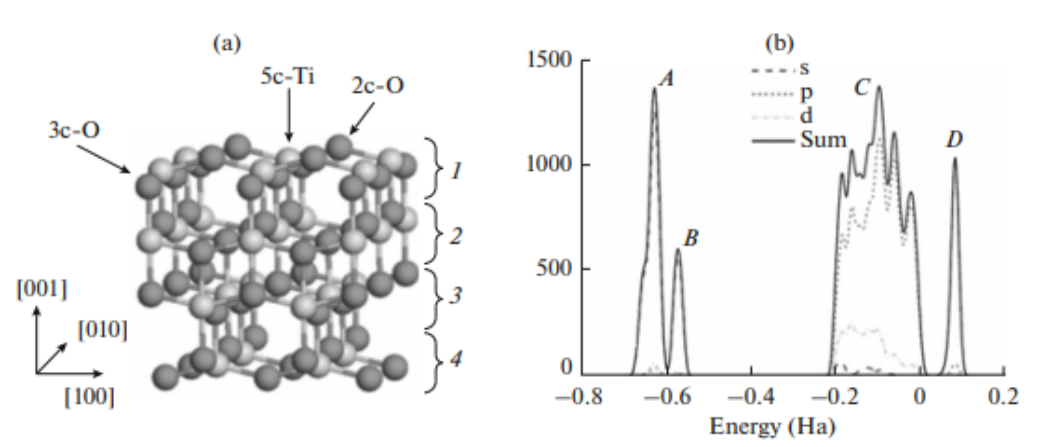
Abstract:
Anodized TiO2 nanotube array (TNA) is a promising material which has attracted wide attentions but its presumed growth mechanism remains enigmatic yet. In this research, Density Functional Theory (DFT) was applied to determine the growth mechanism of TNA on the surface of titanium foil. The firstprinciples within the generalized gradient approximation (GGA) and Perdew-Burke-Emzerhof (PBE) exchange-correlation function based on the density functional theory was employed to calculate anodization process on anatase TiO2 (001) surface. Calculation results indicated that the chain reaction model for multimolecular HF destructive adsorption on surface of TiO2 layer was the key step of anodization to form the initial defects. The HF molecule inclined to adsorb on the defective site owing to the low adsorption energy, resulting in the successive corrosion to deepen the defect and finally to form the nanotube. Complex ion [TiF6]2– can be formed in electrolyte by interaction with 5c-Ti and F- in this corrosion process. This theoretically calculation confirms the growth mechanism hypothesis of TNA.
本工作应用密度泛函理论(DFT)确定了钛箔表面TNA的生长机理,计算了锐钛矿TiO2(001)表面的阳极氧化过程,模拟了钛箔阳极化过程中的关键步骤,即纳米管初始孔的生成。研究了氢氟酸分子在锐钛矿 (001)表面的吸附。计算结果表明,TiO2层表面多分子HF破坏性吸附的链式反应模型是阳极化形成初始缺陷的关键步骤。氢氟酸分子由于吸附能量低而倾向于吸附在缺陷部位,导致连续腐蚀,使缺陷加深,最终形成纳米管。缺陷表面的HF吸附能为0.088 Ha,高于完整表面的0.0768 Ha。在腐蚀过程中,通过与5c-Ti和F-的相互作用,可以在电解液中形成络合物离子[TiF6]2-。这一理论计算证实了TNA的生长机制假说。
Fabrication of hematite nanowire arrays on pure iron via anodization process for superhydrophilic surfaces
Wei Jiang, Jiaping Qiu, Shaojun Yuan, Ying Wan, Jiemin Zhong, and Bin liang
Cite this: Prot Met Phys Chem Surf 51, 435–440 (2015)
https://doi.org/10.1134/S2070205115030107

Abstract:
In this work, hematite nanowire arrays (HNA) were fabricated on pure iron foil by using anodization process in fluoric electrolyte to produce superhydrophilic surfaces. The in situ growth of HNA on iron substrates was ascertained by scanning electron microscopy images, X-ray photoelectron spectroscopy and glancing angle X-ray diffraction measurements. The resultant HNA-bearing iron surfaces showed superhydrophilic property with a contact angle of 7 +/- 2A degrees, and its super-hydrophilicity remained longer than three days in air.
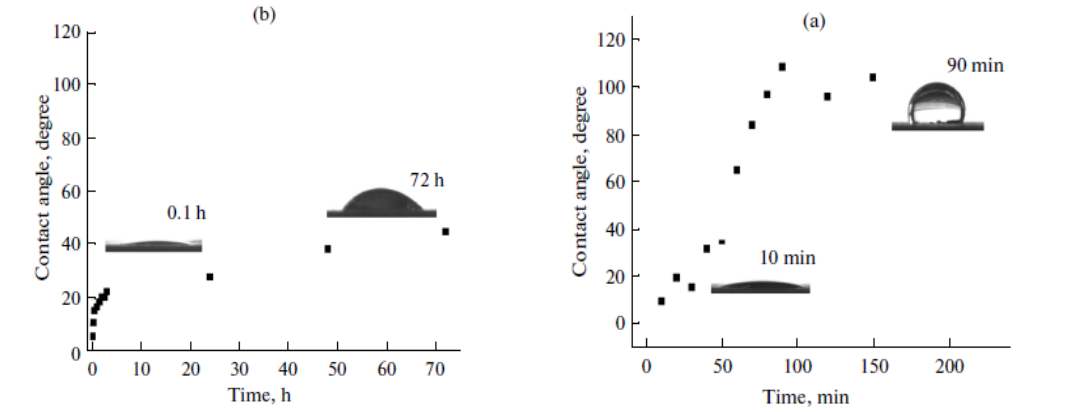
本工作采用氟电解液阳极氧化法制备了具有超亲水表面的赤铁矿纳米线阵列(HNA)。通过扫描电子显微镜、x射线光电子能谱和x射线扫描角衍射等方法,研究了HNA在铁基体上的原位生长。合成的含HNA的铁表面表现出超亲水性,接触角为7 ± 2°,其超亲水性在空气中保持时间超过3天。
Preparation of AgCl Particles with Different Superwettabilities by Particle Size Regulation
Hongyan Fu, Lilin Yang, Yaoguang Wang, Chao Yang, Wen Tian, and Wei Jiang*
Cite this: Langmuir 2019, 35, 7944−7953
https://doi.org/10.1021/acs.langmuir.9b00809
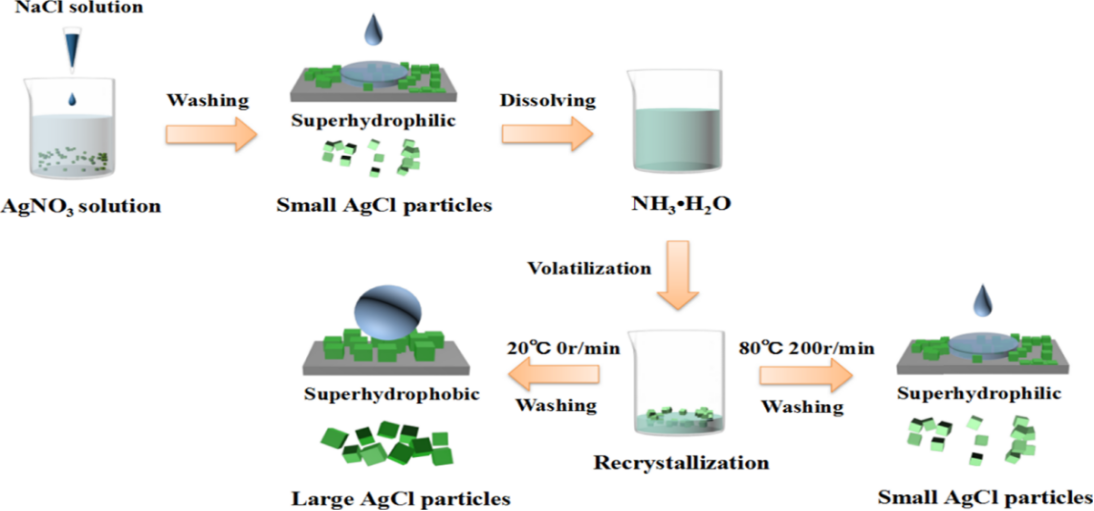
Abstract:
In this work, a simple ammonia evaporation process to obtain AgCl powder with arbitrary superwettability, without introducing any low-surface-free-energy modifier, was investigated. By controlling the recrystallization parameters of the ammonia evaporation process, AgCl crystals precipitated from AgCl−ammonia solution show different wettabilities ranging from superhydrophilicity, via hydrophilicity and hydrophobicity, to superhydrophobicity, with the same chemical composition and structure. Characterization of the obtained AgCl samples with different wettabilities confirms the decisive effect of particle size although light irradiation also causes their wettability transformation
该工作在不引入任何低表面自由能改进剂的情况下,通过控制氨水蒸发过程的再结晶参数,从AgCl-氨水中析出的具有相同结构和成分的AgCl晶体具有不同的润湿性。通过对不同润湿性的AgCl样品的表征证实了粒子尺寸的决定性作用。AgCl润湿性随粒径的均匀变化不依赖于原料成分和结构,不需要低表面能改性剂,为开发绿色、廉价的超疏水颗粒材料制备路线提供了可靠的策略。
Size-dependent superwettability adjustment strategy for preparing superhydrophilic and superhydrophobic solid particles
Wei Jiang*, Hongyan Fu, Lilin Yang, Wen Tian, Houfang Lu, Bin Liang
Cite this: Applied Surface Science 487 (2019) 304–314
https://doi.org/10.1016/j.apsusc.2019.05.081
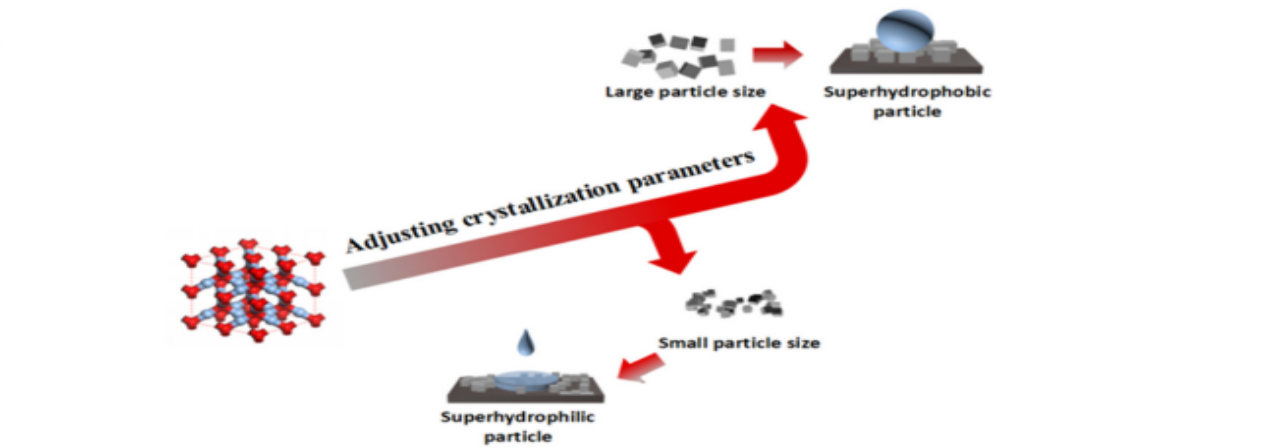
Abstract:
In In this research, a simple strategy to prepare superwetted solid particles based on particle size adjustment is proposed. Typical samples, including Ag2O, AgCl, AgBr, Cu2O, Cu(OH)2, and soot, with different superwettabilities were obtained based on this size-dependent strategy. The acquired superwetted samples without any modification, whether superhydrophilic and superhydrophobic, had the same composition and structure, and their wettability was only decided by the particle size. This size-dependent strategy for preparing superwetted solid particles is universal and promising.
该工作通过对结晶过程的调控,得到了6个典型的超疏水或超亲水样品Ag2O、AgCl、AgBr、Cu2O、Cu(OH)2和煤灰。通过排除了成分、结构和粗糙度的影响后,确定了粒径对超润湿性的决定作用。临界表面能被确定为26mJ /m2,以实现这种超润湿性转变。通过增大或减小粒径来降低或提高表面能以越过该临界值,分别会导致超疏水性或超亲水性的突然转变。这种依赖于尺寸的润湿性机制提供了一种通用的、有前途的策略,可控制地制备超润湿的固体颗粒,在实际应用不需要添加外部改性剂。
Floatable superhydrophobic Ag2O photocatalyst without a modififier and its controllable wettability by particle size adjustment
Wei Jiang*, Hongyan Fu,Yingming Zhu, Hairong Yue, Shaojun Yuan and Bin Liang
Cite this: Nanoscale, 2018, 10, 13661
https://doi.org/10.1039/C8NR02581A
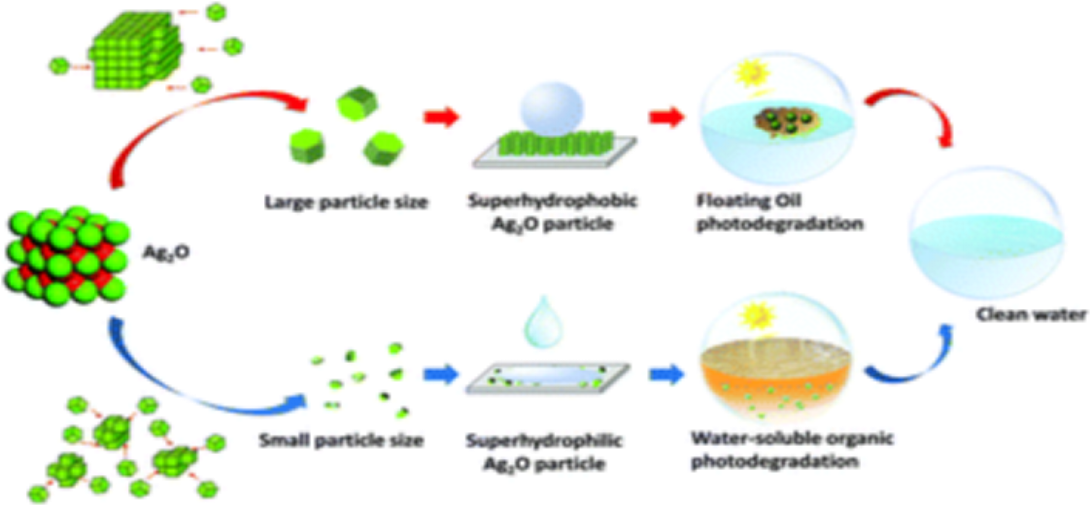
Abstract:
In this research, a controlling mechanism of particle size on the wettability of Ag2O particles is reported, and an interesting floatable Ag2O photocatalyst with superhydrophobicity and superoleophilicity is prepared based on this mechanism. Stable superhydrophobic and superhydrophilic Ag2O without a low-surface-energy modifier can be obtained only by adjusting its particle size, and its wettability can switch mutually by changing the particle size. The wettability of Ag2O converts from superhydrophilic to hydrophobic when the average particle size is more than 1.08 μm. The operation parameters of the Ag2O crystallization process significantly influence the wettability of the Ag2O particles. The obtained superhydrophobic Ag2O floated on the water surface, and exhibited excellent photodegradation performance with various floating oils. This attractive superhydrophobic Ag2O photocatalyst is promising for practical applications, and provides a strategy for the development of functional photocatalysts and superhydrophobic materials.
该研究通过调节结晶过程中晶体尺寸可以制备出具有不同润湿性的Ag2O产物。通过调节溶液浓度、老化时间、研磨时间、添加络合剂类型和浓度等结晶参数,可以实现对Ag2O润湿性的控制和切换。通过调节粒径制备了超疏水性和超亲油性可控的悬浮式Ag2O光催化剂。由于制备的Ag2O光催化剂的超亲油性,对各种浮油均表现出良好的光降解性能。这种特殊的Ag2O材料具有广阔的应用前景,为浮动功能光催化剂和非改性超疏水材料的发展提供了可参考的策略。
Photocatalytic performance of Ag2S under irradiation with visible and near-infrared light and its mechanism of degradation
Wei Jiang,* Zhaomei Wu, Xiaoning Yue, Shaojun Yuan, Houfang Lu and Bin Liang
Cite this:RSC Adv., 2015, 5, 24064
https://doi.org/10.1039/C4RA15774E
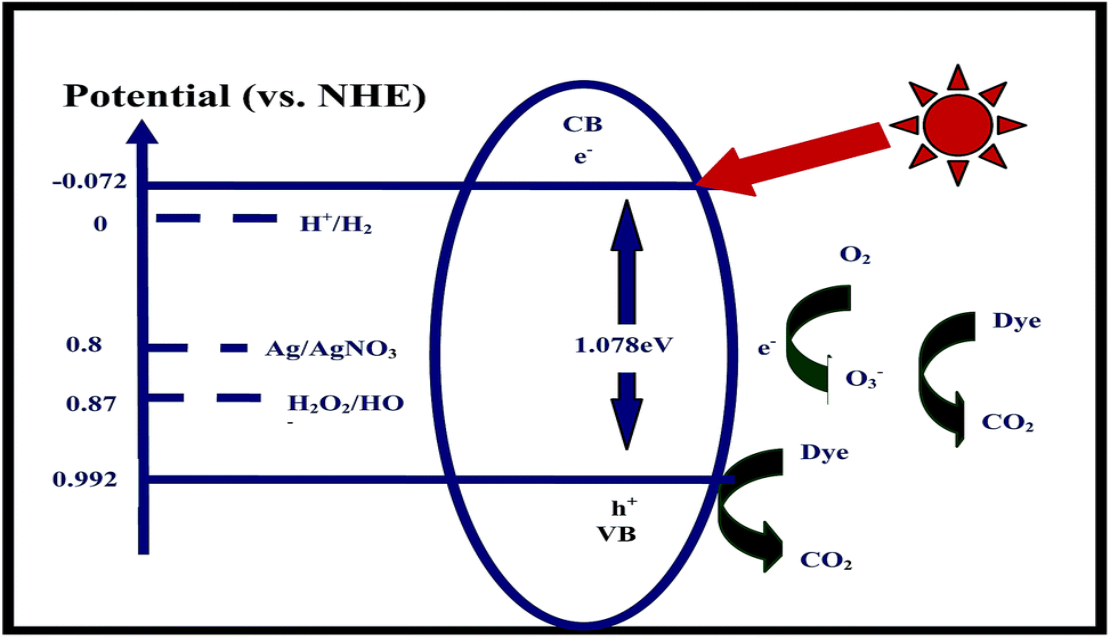
Abstract:
Ag2S has only rarely been investigated as a photocatalyst on its own, although it has been widely used as an important component of composite photocatalysts. We synthesized Ag2S by a facile ion-exchange method at room temperature and used it directly as an effective photocatalyst. Our results confirmed the excellent performance of Ag2S in completely photodegrading methyl orange within 30 min under irradiation with visible light and within 70 min under irradiation with near-infrared light. This good performance is ascribed to the narrow band gap (1.078 eV) of Ag2S and the lower recombination efficiency of the photogenerated electron–hole pairs of Ag2S in the photocatalytic process. The active species in the photo-oxidation process was identified as anioic ozone radicals. This simple preparation method and high photocatalytic performance increases the possible future applications of Ag2S.
采用简单的离子交换法成功地合成了一种Ag2S光催化剂。制备的Ag2S是一种直径为30 ~ 80 nm的晶粒聚集物,是光辐照下MO分解的良好光催化剂。可见光照射下,MO与Ag2S在30min内达到完全降解,近红外光照下在70min内达到完全降解。无论是吸附态还是溶解态,钼在光照射下均被Ag2S完全光降解。这种性能归功于Ag2S (1.078 eV)的窄带隙。
Silver Oxide as Superb and Stable Photocatalyst under Visible and Near-Infrared Light Irradiation and Its Photocatalytic Mechanism
Wei Jiang,* Xiaoyan Wang, Zhaomei Wu, Xiaoning Yue, Shaojun Yuan, Houfang Lu, and Bin Liang
Cite this: Ind. Eng. Chem. Res. 2015, 54, 832−841
https://doi.org/10.1021/ie503241k
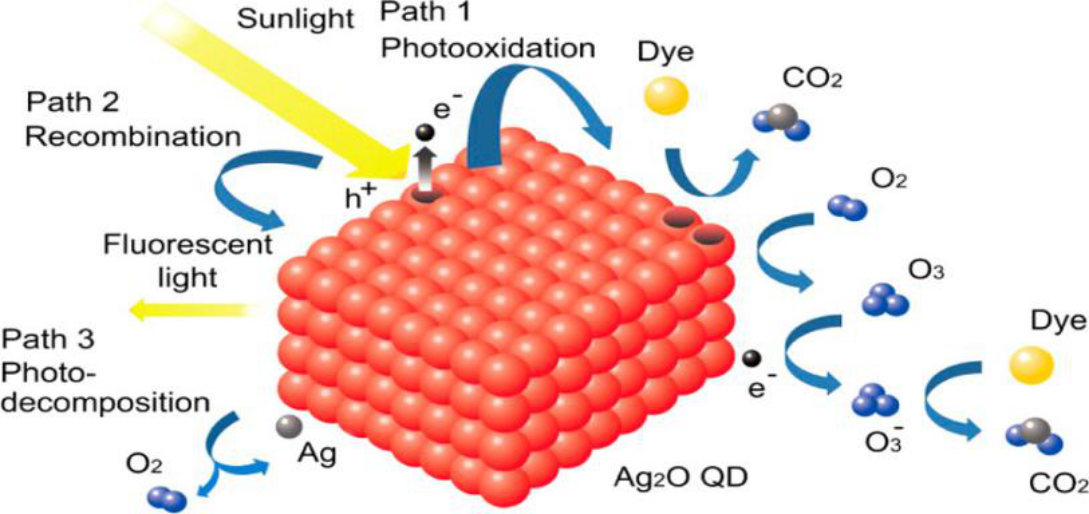
Abstract:
Photocatalytic processes are an environmentally friendly technology for treatment of persistent organic pollutants. However, the majority of current photocatalysts cannot utilize sunlight sufficiently to realize fast decomposition of organic pollutants. In this research, a silver oxide nanoparticle aggregation with superb photocatalytic performance under artificial light source and sunlight was prepared and characterized. The results showed that methyl orange (MO) was decomposed completely in 120 s under irradiation of artificial visible light, artificial ultraviolet light, and sunlight, and in 40 min under near-infrared (NIR) light. The superb photocatalytic performance of as-prepared silver oxide remained almost constant after reuse or exposure under sunlight. It was confirmed that the co-working effect of photogenerated hole and ozone anion radicals did play an important role in the process of MO photodegradation with the existence of Ag2O. The narrow band gap of Ag2O, less than 1.3 eV, resulted in the photocatalytic performance of g2O under NIR light. Furthermore, the high surface area and numerous crystal boundaries provided by the aggregation of Ag2O nanoparticles efficiently increased the escape probability of photogenerated electrons and the contact probability of photogenerated holes with outside materials, assuring superb photocatalytic activity and excellent stability of as-prepared Ag2O samples.
我们成功地制备了新型Ag2O光催化剂,该催化剂在紫外线、可见光和红/近红外光的照射下,采用了一种简便的沉淀方法。在紫外或可见光照射下,甲基橙可以在120秒内被Ag2O完全降解。在红光或近红外光照射下,MO的分解可在2 h内完成,在太阳红光/近红外光照射下可在40 min内完成。Ag2O的这种卓越的光氧化性能在反复使用或暴露在阳光下后几乎保持不变。Ag2O的高效性和稳定性应归功于Ag2O的窄禁带、1.29 eV以及Ag2O QDs提供的高表面积和众多的晶界。不同光照条件下产生的光洞和臭氧阴离子自由基是Ag2O光氧化过程中的活性物质。我们认为这种性能惊人的新型光催化剂具有广阔的应用前景。
Visible-light-driven photocatalytic degradation of non-azo dyes over Ag2O and its acceleration by the addition of an azo dye
Na Bi, Hanxiao Zheng, Yingming Zhu, Wei Jiang ⁎, Bin Liang
Cite this: Journal of Environmental Chemical Engineering. 2018, 6, 3150–3160
https://doi.org/10.1016/j.jece.2018.04.047
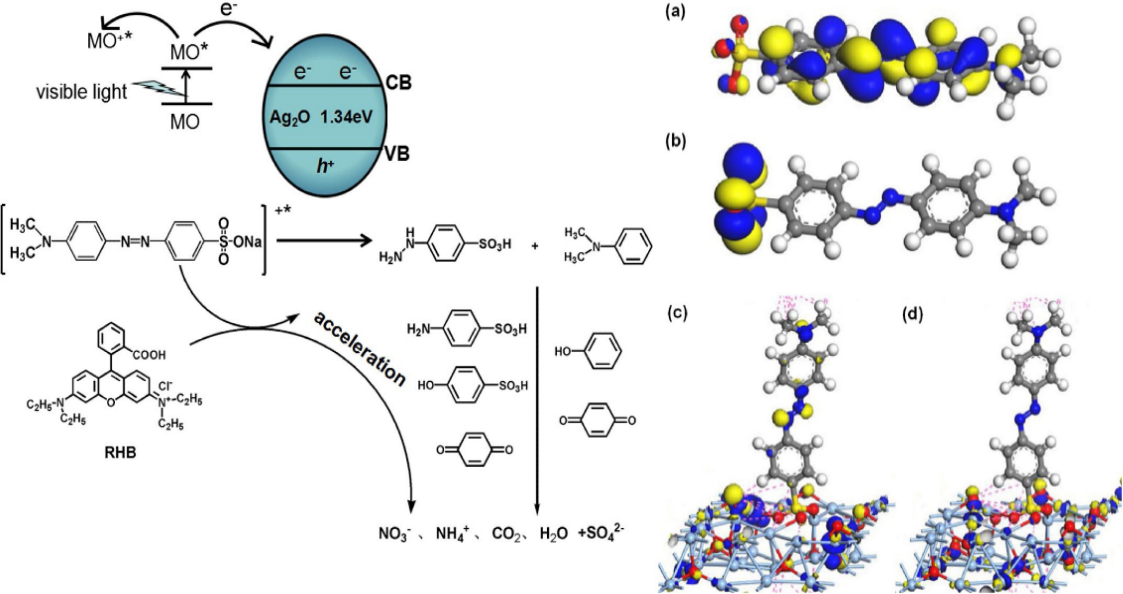
Abstract:
In this study, we reveal that the photodegradation of non-azo dyes using Ag2O as a visible-light-driven photocatalyst is accelerated by the addition of an azo dye. Upon addition of methyl orange, the time taken for the photodegradation of Rhodamine B and methylene blue was significantly shortened from 50 to 18 min and from 20 to 8 min, respectively. This acceleration is not sensitive to the type of azo dye and solution pH. The results indicate that this acceleration can be ascribed to the synergistic effect of the Ag2O and azo species. The intermediates of the photodegradation of the azo species over Ag2O have aniline-, sulfanilic-acid-, and phenol-type structures, and these species promote the photodecomposition of the non-azo organics. This acceleration has promising applications in dye wastewater photodegradation treatment.
在以Ag2O作为可见光驱动的光催化剂时,偶氮染料的加入加速了非偶氮染料的光降解。通过DFT理论计算结合反应过程表征可以证明,本研究发现的光降解加速现象可以归因于Ag2O和偶氮染料的协同作用;偶氮类有机物在Ag2O产生具有苯胺型、磺胺酸型和酚类结构的中间体,这些中间体促进了非偶氮类有机物的光降解。这种协同作用的发现对于Ag2O作为光催化剂用于实际多组分废水的处理具有一定的指导意义,同时也可以促进光催化技术在环境保护方面的应用。
Preparation and Antiscaling Application of Superhydrophobic Anodized CuO Nanowire Surfaces
Wei Jiang,* Jian He, Feng Xiao, Shaojun Yuan, Houfang Lu, and Bin Liang
Cite this: Ind. Eng. Chem. Res. 2015, 54, 6874−6883
https://pubs.acs.org/doi/abs/10.1021/acs.iecr.5b00444
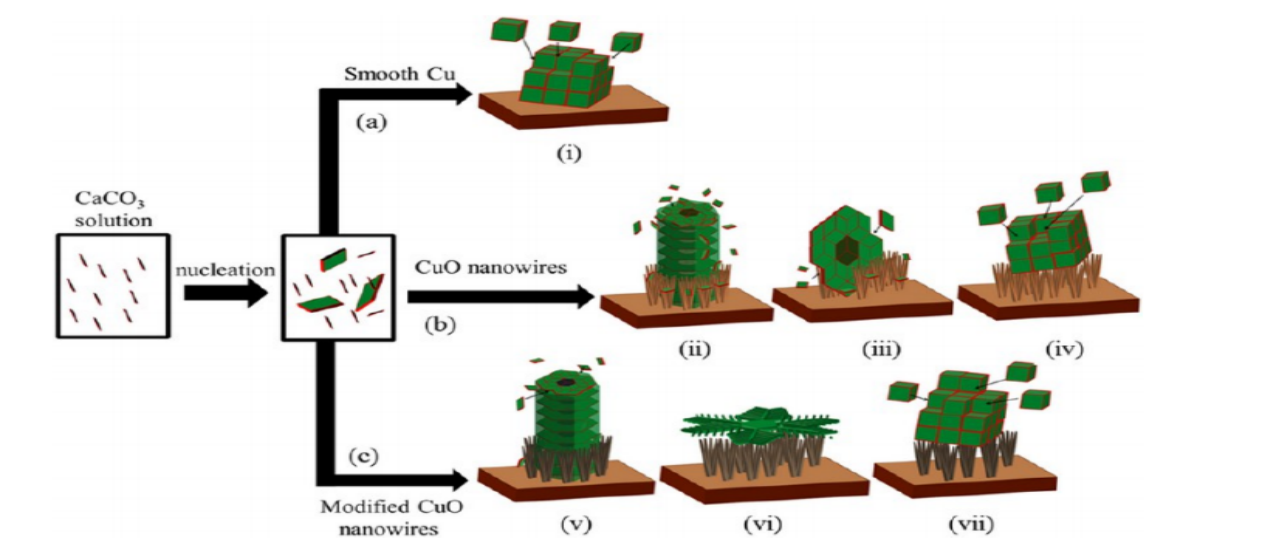
Abstract:
Antiscaling technology is necessary in order to prevent the performance loss and blockage of heat exchangers. In this research, a superhydrophobic CuO nanowire layer was prepared and utilized for antiscaling process of CaCO3 on the surface of copper. Modified with 1H,1H,2H,2H-perflfluorodecyltriethoxysilane (FAS-17), the water contact angle on the CuO surface increased sharply from 4.5° ± 1° after anodization to 154° ± 2°, since the surface free energy decreased from 74.8 mJ/m2 for the hydrophilic surface to 0.2 mJ/2 for the superhydrophobic surface. The scale inhibition performance of the surface of superhydrophobic CuO nanowires was confirmed since the corresponding scaling weight of deposited CaCO3 decreased significantly from 0.6322 mg/cm2 to 0.1607 mg/cm2. This attractive antiscaling effect of the modified superhydrophobic CuO nanowire surface should ascribe to the slow CaCO3 crystal nucleation rate, because of the low surface energy, low adhesion strength of CaCO3 crystal, and air film retained on the superhydrophobic surface.
本研究制备了超疏水CuO纳米线层用于CaCO3的阻垢过程。通过用1H、1H、2H、2H-全氟癸基三乙氧基硅烷(FAS-17)改性,CuO表面自由能从74.8 mJ/m2下降到0.2 mJ/m2,实现了接触角从4.5°±1°急剧增加到154°±2°。CaCO3相应的结垢重量从0.6322 mg/cm2减小到0.1607 mg/cm2,证实超疏水氧化铜具有良好的阻垢效果
Recyclable CoFe2O4-Ag2O magnetic photocatalyst and its visible light-driven photocatalytic performance
Qihui Zeng1 • Yingming Zhu1 • Wen Tian1 • Wei Jiang1 • Bin Liang1
Cite this:Res Chem Intermed (2017) 43:4487–4502
https://doi.org/10.1007/s11164-017-2891-x
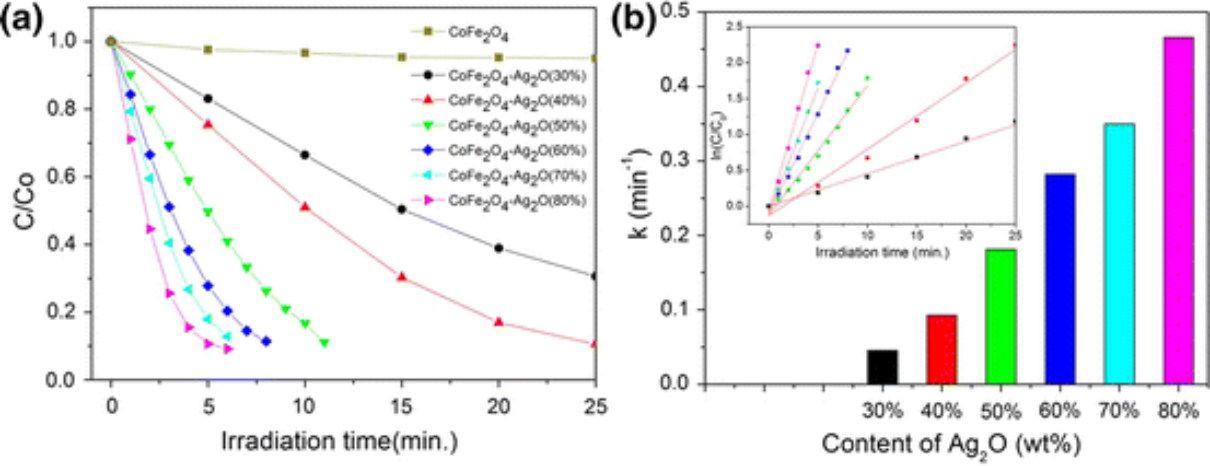
Abstract:
A magnetically separable photocatalyst, the composite compound CoFe2O4–Ag2O, was fabricated successfully by a simple precipitation method and used for photodegradation of organic pollutants under visible light (>420 nm) irradiation. Its magnetic separation performance was evaluated. Results showed that the CoFe2O4–Ag2O with 60-wt% content of Ag2O had the best photocatalytic performance, stability, and magnetic separation performance. Methyl orange, methylene blue, rhodamine B, and phenol can be completely photodegraded by the CoFe2O4–Ag2O photocatalyst in a short period. After five cycles, CoFe2O4–Ag2O kept its performance stability. As prepared, CoFe2O4–Ag2O (60%) has a coercivity of 2500 Oe and a saturation magnetization of 22.45 emu g−1; it can be completely separated magnetically in 20 s with magnetic recovery mass rate of 85% under an external magnetic field. This superb photocatalytic performance and separation recovery confirms that the CoFe2O4–Ag2O photocatalyst is a promising candidate for future use in photo-oxidative degradation of organic contaminants.
本研究制备了一系列不同Ag2O含量的磁分离CoFe2O4-Ag2O杂化光催化剂并对其进行了评价。CoFe2O4 - Ag2O杂化物在可见光和近红外光照射下表现出优异的光催化性能。优化后的混合CoFe2O4 - Ag2O(60%)在120 min内对MO、MB、RhB、苯酚等典型的持久性有机物进行了完全光分解,该混合光催化剂可以进行有效的磁分离,且无较大的质量损失,可以重复使用,且性能没有明显的下降。这种优异的光催化性能和分离回收率证实了CoFe2O4-Ag2O光催化剂是一种很有前途的光氧化降解有机污染物的化合物。
Systematic research on Ag2X (X = O, S, Se, Te) as visible and near-infrared light driven photocatalysts and effects of their electronic structures
Wei Jianga,∗,Zhaomei Wua,Yingming Zhub,Wen Tiana,Bin Lianga
Cite this: Applied Surface Science 427 (2018) 1202-1216
https://doi.org/10.1016/j.apsusc.2017.08.053
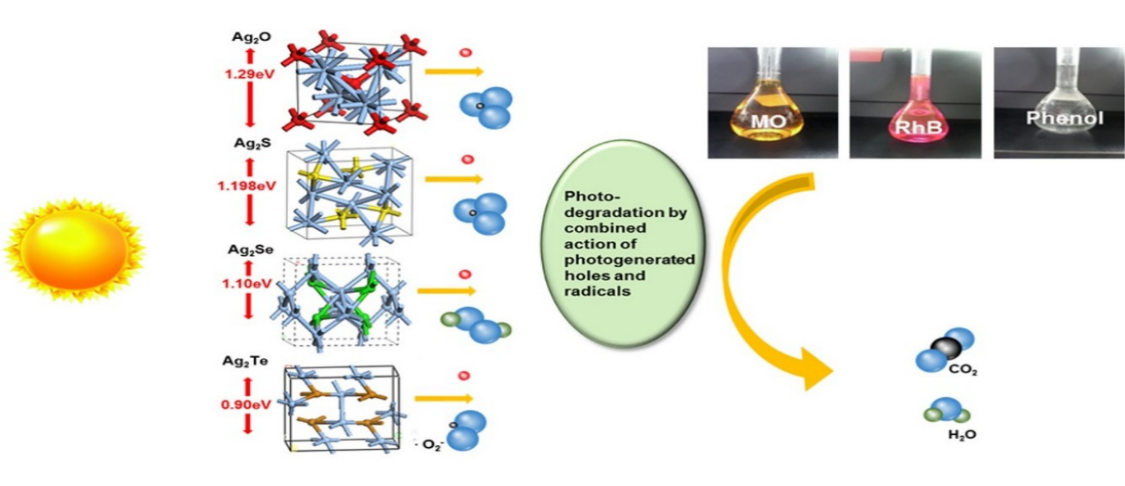
Abstract:
Four silver chalcogen compounds, Ag2O, Ag2S, Ag2Se and Ag2Te, can be utilized as visible-light-driven photocatalysts. In this research, the electronic structures of these compounds were analyzed by simulation and experiments to systematically reveal the relationship between photocatalytic performance and energetic structure. All four chalcogenides exhibited interesting photocatalytic activities under ultraviolet, visible and near-infrared light. However, their photocatalytic performances and stability significantly depended on the band gap width, and the valence band and conduct band position, which was determined by their composition. Increasing the X atomic number from O to Te resulted in the upward movement of the valence band top and the conduct band bottom, which resulted in narrower band gaps, a wider absorption spectrum, a weaker photo-oxidization capacity, a higher recombination probability of hole and electron pairs, lower quantum efficiency, and worse stability. Among them, Ag2O has the highest photocatalytic performance and stability due to its widest band gap and lowest position of VB and CB. The combined action of photogenerated holes and different radicals, depending on the different electronic structures, including anion ozone radical, hydroxide radical, and superoxide radical, was observed and understood. The results of experimental observations and simulations of the four silver chalcogen compounds suggested that a proper electronic structure is necessary to obtain a balance between photocatalytic performance and absorbable light region in the development of new photocatalysts.
本研究模拟合成了四种银硫化合物,研究了它们的光物理和光催化性能。电子结构分析表明,银化合物的光催化性能和稳定性与它们的带隙和位置有关,这取决于它们的组成。随着X原子序数的增加,从O到Te, VB顶部和CB底部的位置向上移动,导致带隙更窄,光氧化能力更弱,空穴和电子对复合概率更高,量子效率更低,稳定性更差。VB和CB在Ag2O中最宽的带隙和最低的位置导致了最高的光催化性能和稳定性,提供了一个在足够宽的光谱下运行的有希望的光催化剂。然而,这四种银化合物都能在紫外线、可见光和近红外光的照射下工作,这是因为它们的带隙相对较窄。光生空穴和自由基,如阴离子臭氧自由基、氢氧自由基、超氧化物自由基等的联合作用可以通过它们不同的能量结构来观察和理解。实验和仿真结果表明,光催化剂必须具有合适的含能结构。太窄的带隙导致光催化性能急剧下降,尽管宽的光区可以利用。对具有相同家族元素的化合物进行系统的研究,可以更深入地了解光催化过程,为开发高性能的光催化剂提供可参考的策略。
Preparation of Silver Carbonate and its Application as Visible Light-driven Photocatalyst Without Sacrificial Reagent
Wei Jiang*, Ya Zeng, Xiaoyan Wang, Xiaoning Yue, Shaojun Yuan, Houfang Lu and Bin Liang
Cite this: Photochemistry and Photobiology, 2015, 91: 1315–1323
https://doi.org/10.1111/php.12495|

Abstract:
Visible light‐driven photocatalyst is the current research focus and silver oxyacid salts with p‐block elements are the promising candidates. In this research, Ag2CO3 was prepared by a facile precipitation method and used to degrade the pollutants from waters. The results revealed that the silver carbonate with monoclinic structure quickly decomposed methyl orange and rhodamine B in less than 15 min under visible light irradiation. When it was recycled six times, the degradation of methyl orange still can reach 87% after 30 min. The calculated band gap of Ag2CO3 was 2.312 eV with Valence band edge potential of 2.685 eV and Conduction band 0.373 eV vs NHE, which endowed the excellent photo‐oxidation ability of silver carbonate. Photogenerated holes and ozone anion radicals were the primary active species in the photo‐oxidization degradation of dye. The generation of metallic silver resulted from photocorrosion and the consequent reduction in the ozone anion radical amount led to the performance degradation of Ag2CO3. The simple preparation method and high photocatalytic performance of Ag2CO3 increases its prospect of application in future.
一种新型的可见光响应型光催化剂Ag2CO3通过简单的沉淀法成功制备。其带隙为2.312 eV,计算VB和CB边缘电位分别为2.685和0.373 eV。MO和RhB的光降解实验表明,在不添加任何牺牲试剂的情况下,制备的Ag2CO3具有良好的光氧化性能和稳定性。首次使用时,98%的MO分解不超过15min,循环6次后,30min后MO的降解率也可达到87%。



